While the animal research industry continues to breed, buy, cage, torture, and kill sentient beings, progressive scientists are busy proving that human-relevant science is not only possible but, in fact, better for us all. Highlights in science from the last month are below.
Organoids Drive Human-Relevant Drug Discovery
Genetic Engineering & Biotechnology News, 4/1/2026
“Drug discovery increasingly relies on three-dimensional (3D) cell models to address a persistent challenge in biomedical research: traditional experimental systems often fail to accurately reflect human biology. Organoids, spheroids, and other complex cellular models allow scientists to recreate aspects of tissue structure and function in the laboratory. These systems now play a central role in new approach methodologies (NAMs), experimental strategies designed to produce human-relevant biological data while reducing reliance on animal testing.”
“‘With historical models we often lack biological relevancy, and animal models don’t always recapitulate human biology the way we want.’ That disconnect between animal studies and human biology has long slowed drug development. Compounds that perform well in animals frequently fail during clinical trials, forcing researchers to spend additional time and resources identifying therapies that actually work in people. Three-dimensional cell models aim to narrow that gap by recreating tissue-like environments where cells interact with neighboring cells and the surrounding extracellular matrix.” 📰 Full Story →
Loughborough spin-out secures £325k to accelerate drug testing with lab-grown human muscles
Loughborough University, 4/2/2026
“A Loughborough University spin-out that aims to revolutionise drug testing using lab-grown human muscles has been given a significant funding boost. Myomaker Bio has received £325,000 investment from SFC Capital to help its mission to speed up drug development and bring new treatments to market.”
“The team of scientists and researchers behind the business are world-leaders in muscle biology and have developed human muscle organs and tissues that closely behave like real muscles in the human body. By using these muscles to test drugs, Myomaker Bio aims to speed up the creation of new medicines for rare diseases and more accurately predict what will happen in real patients before moving to clinical trials. It also hopes this will cut the use of animal testing.” 📰 Full Story →
Accelerating Organoid Adoption Set to Transform Cancer Drugs
Fay Lin, Genetic Engineering & Biotechnology News, 4/2/2026
“In cancer research, organoid biobanks that capture diverse genetic backgrounds, ethnicities, ages, and other patient-specific variables are opening new avenues for precision medicine. Yet Hans Clevers, MD, PhD, renowned stem cell biologist and the ‘father of organoids,’ explains that replacing decades of infrastructure reliant on animal models is not an easy feat. ‘In Dutch, we call this koudwatervrees—cold water fear,’ said Clevers during his keynote session at GEN’s Spotlight on Organoids….”
“When asked about the biggest obstacles preventing organoids from reaching their full potential, Alif Saleh, CEO of 28bio, emphasizes that adoption, not technology, is the primary hurdle, particularly in drug discovery, where a staggering 95% failure rate is accepted as the norm. . . . He states that achieving a large cultural shift for widespread organoid adoption will likely require either regulatory pressure or an organoid-powered commercial success.”
“‘Have you ever bet $3 billion on a single number in roulette? That’s essentially drug discovery,’ said Robert DiFazio, PhD, co-founder and co-CEO at Parallel Bio, in response to pharma’s high failure rates. ‘Pharma needs to recognize that animal testing is a problem.’” 📰 Full Story →
Human-Relevant Models Come of Age
The Medicine Maker, 4/6/2026
“‘To a great extent, this is a case of legislation catching up to the science. There was already significant belief in the efficacy of human-relevant models. However, uncertainty around regulatory acceptance and standards had an understandable effect on innovation and adoption.’”
“‘Late-stage failure is often caused by one of two major factors. Either the drug does not work in humans in precisely the way it does in animals, or an adverse effect emerges that was not predicted in testing. Human-relevant models respond to these issues by enabling teams to anchor decisions in human biology earlier. This is particularly vital in immune signaling, receptor pharmacology and some vascular liabilities, where differences between species can prove problematic in testing. These models can identify mismatches at an early stage. If you discover that you cannot achieve the penetration necessary for drug efficacy, or you encounter safety signals under human-relevant exposure conditions, you can course-correct at that point, rather than discovering these issues after investing in clinical readiness and execution.’” 📰 Full Story →
“Cervix-on-a-chip” to help discover new STI treatments
University of Maryland School of Medicine, healthcare-in-europe.com, 4/8/2026
“Sexually transmitted infections (STIs) not only impact an individual’s health but also result in multibillion-dollar economic losses worldwide. To study these diseases, a team of researchers has developed the first-of-its-kind immune-capable ‘organ-on-a-chip’ model that realistically reproduces the human cervical environment, allowing scientists to study how the microbiome, immune system, and STIs interact—something that has not been possible before with oversimplified cell cultures or animal models.”
“‘This new model will revolutionize how scientists study STIs, leading to an improved understanding of these conditions, as well as the potential for better treatments,’ said co-lead author Jacques Ravel, PhD, Director of the Center for Microbiome Research and Innovation (CAMRI) within UMSOM’s Institute for Genome Sciences (IGS); the John L. Whitehurst Professor of Medicine, Microbiology and Immunology and Assistant Dean for Research Advancement at UMSOM. ‘The other powerful part of this research has been its cross-discipline collaboration in the research. By integrating engineering, microbiology, immunology, and microbiome science, we were able to build a model that more closely reflects human biology and the complexity of the cervical microenvironment.’”
“‘For the first time, we can simulate what happens in the human body rather than relying solely on a petri dish systems or inadequate animal models.’” 📰 Full Story →
Human skin model to help treat skin stiffened by age and disease
The University of Western Australia, 4/8/2026
“Researchers from The University of Western Australia will use biomaterials to create a lifelike 3D model of human skin to better understand and treat skin stiffness associated with age, scarring and disease. If successful, in time the model could reduce the need for animal testing of skin products and enable personalised treatment plans for patients experiencing abnormal skin stiffness.”
“‘Significantly, by developing a realistic, full-thickness human skin tissue model, it could potentially reduce animal use in research and product development.’” 📰 Full Story →
Researchers create a new human derived adipose tissue model that helps to understand obesity without animal testing
Tampere University, 4/9/2026
“Research on obesity and associated diseases still relies on animal models, which do not accurately reflect human physiology. The three-dimensional human-based adipose tissue model developed by doctoral researcher Sini Saarimaa and colleagues at Tampere University reveals interactions between the nervous system and adipose tissue and may shed new light on the mechanisms of obesity.”
“The developing tissue model may help to identify new drug molecules that act directly on adipose tissue. It can also help reduce the use of animal models, as it provides a more relevant, human-based research platform that supports the development of better prevention and treatment methods. At the same time, it advances personalised medicine in the treatment of obesity and related metabolic diseases.” 📰 Full Story →
Israeli researcher advances 3D bioprinting toward organ transplants, ‘opening door to better solutions’
Eitan Gefen, ynetglobal, 4/12/2026
“The idea of producing parts of the human body in a lab may sound like science fiction, but it is already happening. While full organs such as hearts or kidneys are not yet within reach, Israeli researchers see their work as a meaningful step toward that goal. At Rambam, scientists are working to print human tissues using a patient’s own cells, create disease models and test new drugs before they reach human trials.”
“If the technology reaches its full potential, the implications are profound. Today, there is a severe shortage of organs for transplant, and patients often wait years. In the future, researchers hope it will be possible to take cells from a patient and print a personalized organ, eliminating the risk of immune rejection.”
“For now, the most immediate applications lie in drug development. ‘One of the biggest challenges in developing drugs is toxicity,’ Artzy-Schnirman said. ‘A drug may solve one problem, but harm the liver or heart. There is currently no simple way to test this.’ Bioprinting allows researchers to create human tissue models, such as liver or heart tissue, and test drugs on them. . . . The technology could also reduce reliance on animal testing.” 📰 Full Story →
CU Denver Creates Lab-Grown Lung Model to Test Treatments More Effectively
CU Denver News, 4/13/2026
“University of Colorado Denver (CU Denver) Biomedical Engineering Associate Professor Chelsea Magin is developing a lung model to help advance the treatment of lung diseases which affect men and women differently. The lung is made by combining donor cells and tissues with synthetic materials. The result behaves like a real lung—soft when healthy, stiff when sickly—allowing researchers to study diseases like pulmonary fibrosis and cancer more accurately.”
“Current methods fall short. Many lab-grown cells are studied on flat, rigid surfaces that don’t reflect how lungs function. Animal testing also has limits. As a result, nearly 90% of drugs that work in the lab fail in human trials. Magin’s model aims to close that gap.” 📰 Full Story →
How Frontier Bio Plans to End the Organ Shortage
causeartist, 4/17/2026
“Every 10 minutes, someone in the U.S. is added to an organ transplant waitlist. Thirteen people die each day waiting for a donor that may never come. Only about 10 percent of global organ demand is currently met. . . . Eric Bennett thinks the field has been solving the wrong subproblem. The organ shortage isn’t primarily a donor matching problem or an immune suppression problem. It’s a manufacturing problem. And manufacturing problems are solvable.”
“The answer Frontier Bio is building toward: living blood vessels that replace synthetic grafts, integrate seamlessly with the patient’s own body, and eventually become the vascular infrastructure for fully engineered organs. . . . The scale of potential impact made it clear: we have a chance to drastically reduce reliance on animal testing and eventually eliminate the organ transplant waitlist entirely.” 📰 Full Story →
Organ-Chips Accelerate Smarter Drug Decisions
Emulate and The Scientist, The Scientist, 4/19/2026
“Emulate reported that the human Liver-Chip correctly identified 87 percent of small molecule drugs known to cause human [drug-induced liver injury (DILI)], with 100 percent specificity, across a blinded set of 27 drugs tested using 870 chips. Because each of these drugs had been in the clinic, they must have been tested on an animal. Thus, by extension, if the animal did identify a risk, it did not affect the decision to progress to the clinic. Of the drugs tested, Emulate was able to find public reports that totaled nearly 250 patient fatalities, which calls into question the ability of the animal to correctly predict the risk. It is known that there are species differences in metabolism and drug transporters, both of which are important mechanisms leading to DILI. Thus, using human cells in a physiologically appropriate microenvironment has led to the superiority of the Liver-Chip compared to the animal models within the drug set that was tested.” 📰 Full Story →
Norway says “enough” to animal testing. A new center aims to change the entire research system
MyNorway, 4/22/2026
“The Norwegian government has announced the establishment of a national 3R center. Its goal is to develop alternatives to animal testing. The decision was announced by the Ministry of Agriculture and Food on April 17. A budget of NOK 6 million has been allocated for 2026. This is the result of over 20 years of efforts by Dyrevernalliansen (the Animal Protection Alliance).”
“Dyrevernalliansen points out that this is a breakthrough after years of advocacy. The organization expects further steps and calls for the creation of a national plan to phase out animal use in research. . . . It also highlights the need for increased funding and prioritization of alternatives. . . . ‘It is high time for Norway to also abandon animal testing and switch to safer and more ethical methods wherever possible.’” 📰 Full Story →
Advanced Immune-Integrated Models Pave the Way for Next-Generation Wound Healing Research
Scienmag, 4/23/2026
“In the quest to unravel the complexities underlying impaired wound healing and pathological scarring, researchers are turning to innovative next-generation models that seamlessly integrate the immune system’s pivotal role. . . . Organ-on-a-chip technologies emerge as particularly transformative for modeling complex immune-stromal interactions under physiologically relevant conditions. . . . Hybrid approaches that combine organoids, 3D bioprinting, and modular microfluidics push the envelope further by enhancing structural complexity and functional realism.”
“[] the impact of immune-integrated wound models on translational science
is poised to be profound. By bridging the gap between in vitro experimentation and in vivo relevance, these platforms transcend the limitations of animal models, which often poorly predict human immune responses due to interspecies differences. Consequently, pharmaceutical development pipelines stand to benefit from more predictive drug screening environments, enabling early identification of efficacious compounds and mitigating late-stage failures. Moreover, these technologies lay the groundwork for precision medicine approaches where patient-derived immune cells inform individualized treatment strategies.” 📰 Full Story →
The Organ-on-Chip, Microphysiological Systems Landscape, NAMS: A Business Overview
R.D. Solorzano, The Business of Biofabrication, 4/17/2026
“The Organ-on-Chip (OoC), Microfluidics, and New Approach Methodologies (NAMs) space is no longer a niche academic pursuit, it is rapidly becoming a commercial force reshaping how drugs are discovered, toxicology is assessed, and disease is modelled. What was once a handful of pioneering labs has expanded into a dense, competitive ecosystem of startups, scale-ups, and platform companies, each staking out distinct territory across biology, engineering, and data science.”
“For investors, pharma partners, and drug developers, the question is no longer whether this technology works, it’s which platforms are positioned to win, and where the commercial opportunity is greatest. This article maps the emerging landscape across key segments, highlighting the companies and leaders driving the space forward.” 📰 Full Story →
VivoSim Platform Predicts Gastrointestinal Toxicity (Diarrhea) in an AI Model Trained on Human Intestinal Model Results – Without Animal Testing
Globe Newswire, Business Insider, 4/23/2026
“VivoSim Labs, Inc. (Nasdaq: VIVS) (the “Company” or “VivoSim”), a provider of next-generation New Approach Methodologies (NAMs) for preclinical safety, today announced the availability of an AI prediction tool leveraging its NAMkind™ intestinal models to accurately predict the potential of a given drug compound to cause diarrhea in patients. The tool integrates its proprietary NAMkind™ ileum and colon tissues with advanced machine‑learning analytics to identify drug‑induced disruptions to intestinal epithelial integrity and function. Tissue-based assay data is used to train an AI predictive model in a process VivoSim has named VitroSense™.”
“The model was built using a training set of dozens of compounds. Using high-quality real-world 3d NAM assay results generated from the set of training compounds, the model attained a predictive accuracy of 96% for potential diarrhea. This success represents a significant step forward for the field, demonstrating the value of combining data from VivoSim’s complex 3D cell-based assays with machine learning approaches.” 📰 Full Story →
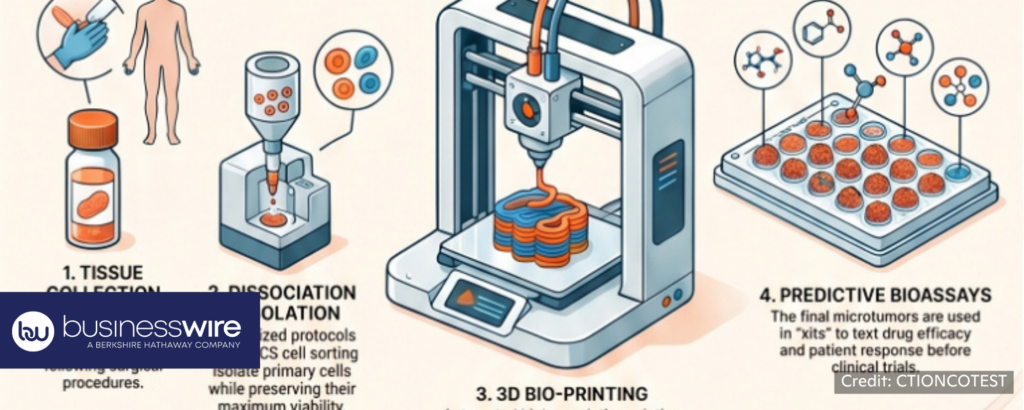
Printing the Future of Oncology: CTIBIOTECH Invests €3M and Secures €1.25M from France 2030 to Revolutionize Personalized Cancer Care
businesswire, 4/28/2026
“CTIBIOTECH™, an innovative French Contract Research, Development, and Manufacturing Organization (CRDMO) based in Meyzieu-Lyon, proudly announces the launch of the CTIONCOTEST ™ project, a groundbreaking €3 million initiative aimed at transforming preclinical cancer research. To accelerate this critical innovation, CTIBIOTECH ™ has been awarded €1.25 million in funding from Bpifrance under the “Aide au Développement Deeptech” program, with the strategic support of France 2030 and the Government of France.”
“Currently, 95% of new cancer drugs fail during human clinical trials, costing the pharmaceutical industry billions of euros and delaying life-saving treatments for patients. The CTIONCOTEST ™ project addresses this urgent global health challenge by developing an automated, industrial-scale process to 3D bioprint hundreds of standardized human “microtumors” directly from a specific cancer patient’s primary tumor cells.”
“By providing a highly predictive, 100% human-derived testing platform, CTIONCOTEST ™ actively supports the regulatory shift towards New Approach Methodologies (NAMs), drastically reducing and replacing the need for animal experimentation in preclinical trials.” 📰 Full Story →
Post and Courier ‘Monkey Business’ story wins national Headliner Award
The Post and Courier, 4/28/2026
“The Post and Courier’s watchdog work on the controversial monkey breeding colony in Yemassee and on South Carolina’s Morgan Island earned a first place National Headliner Award. Written and reported by Marilyn W. Thompson and Mitchell Black, the stories explored a research center in Yemassee, which received international attention after 43 rhesus monkeys staged a breakout.”
“The National Headliner judges said of Thompson’s and Black’s work: ‘Incredible in-depth reporting on the history and current controversy over monkey breeding for medical testing in South Carolina. Pieces were well-written, well-researched, and the team clearly spent a lot of time on or near the island verifying claims and doing the work.’” 📰 Full Story →
Why the president of an Immokalee primate importer was charged with a misdemeanor
Cary Barbor, WGCU NEWS, 4/28/2026
“The president of a local company that imports primates was charged with a misdemeanor involving mistreatment of animals earlier this year. BC US is an Immokalee company that breeds, quarantines, and sells macaque monkeys for research and testing.”
“This spring, the Florida Fish and Wildlife Conservation Commission charged the company’s president, Mark Bushmitz, with a second-degree misdemeanor for the escape of Class 2 wildlife. The charge carries a penalty of 60 days in jail and/or a fine of up to $500. Charging a company officer for animal mistreatment is extremely rare.” 📰 Full Story →
U of T researchers improve maturity of lab-grown heart cells for disease modelling
Qin Dai, University of Toronto Faculty of Applied Science & Engineering, 4/29/2026
“Researchers at the University of Toronto’s Institute of Biomedical Engineering have developed a new method to mature lab-grown heart cells, so they more closely resemble adult human heart tissue. By optimizing the chemical cocktail in which these cells are grown, the team improved their structure, electrical activity and ability to contract. This advance could help create more reliable models for studying heart disease and testing new drugs, where current lab-grown cells often fall short due to their immature state.”
“‘Animal models often aren’t very reliable in predicting this cardiotoxicity, and as a result it is slower and more expensive to produce effective drugs. Ultimately, patients are left waiting. We hope that these new methods can help to improve our predictions of cardiotoxicity in pharmaceutical testing.’” 📰 Full Story →
Bern researchers develop artificial placenta to test medicine
swissinfo.ch, 4/30/2026
“It is unclear for most drugs whether and to what extent they are passed on to an unborn child . . . Many drugs are therefore not authorised for pregnant women as a precautionary measure, even though they may not pose a risk. Animal studies are also not always meaningful for these questions, as the placenta – the barrier that protects the foetus [sic] from toxins – functions very differently depending on the animal.”
“To remedy this, a team at the University of Bern led by biomedical scientist Christiane Albrecht has now developed a model in an [Swiss National Science Foundation (SNSF)]-funded project that is intended to better represent the transport of substances in the human placenta than previous methods. . . . In future, the researchers want to use the model to investigate not only the path of drugs, but also the transport of substances such as iron and cholesterol. Such models could also help to reduce the number of animal experiments.” 📰 Full Story →
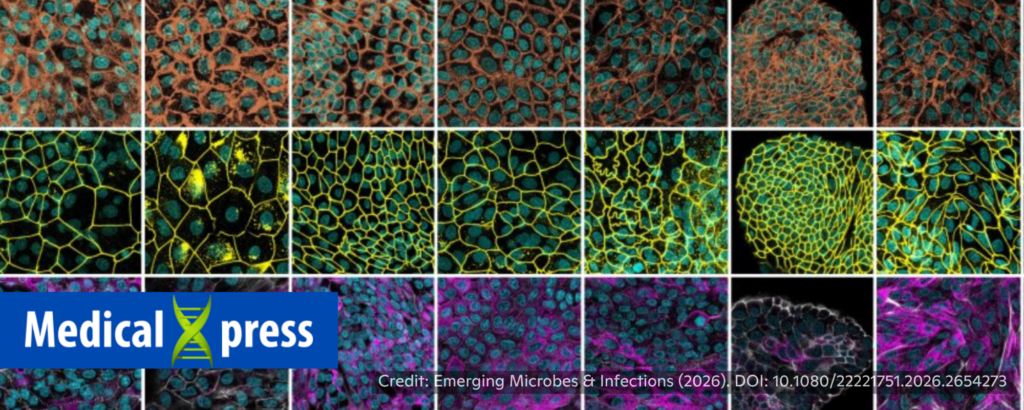
No live animal testing needed: Lab-grown airway organoids reveal viral infection in wildlife species
Sanjukta Mondal, Medical Xpress, 4/30/2026
“Experimental infections, where a pathogen is introduced into the host body to see its effects in action, are considered the gold standard for assessing how vulnerable a host is, offering clear insights into how a pathogen causes disease and spreads through populations. This approach requires deliberately infecting live animals, which not only raises clear ethical concerns but also often proves impractical for many wildlife species, especially endangered ones, where conservation efforts take precedence. Scientists have found a potential solution to this problem. ”
“They developed a new method for growing miniature organs, called airway organoids, in the lab for 10 different animals, including wildlife such as the red panda and Goeldi’s monkey, as well as livestock. They turned tissue samples from each animal into 3D airway models that recreate how the respiratory tract functions and is built at the cellular level, enabling researchers to test susceptibility to influenza viruses of both mammalian and avian origin. Tests conducted using the organoids showed large differences in infection trends across animal species and in the extent of damage the virus causes in their cells. These results were consistent with what is already known from real-life infections and field studies.” 📰 Full Story →
When you share these breakthroughs, you’re helping expose a truth the animal research industry wants to hide: ethical science is better for everyone.
Share this science news roundup on Facebook, X, or Bluesky now.