While the animal research industry continues to breed, buy, cage, torture, and kill sentient beings, progressive scientists are busy proving that human-relevant science is not only possible but, in fact, better for us all. Highlights in science from the last month are below.
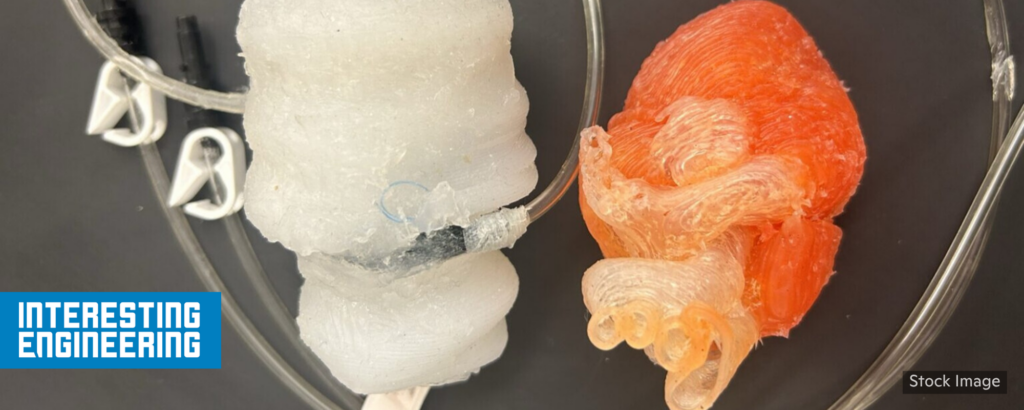
3D-printed ‘beating heart’ offers new frontier simulations
Munis Raza, Interesting Engineering, 3/5/2026
“Heart disease remains a primary cause of illness and mortality around the world. While minimally invasive procedures have improved cardiac care, the heart’s complex anatomy and constant motion require rigorous hands-on training to reduce procedural errors. To address this issue, researchers have developed a 3D-printed dynamic heart model that mimics the complex movements of the left side of the heart. This model includes atrium, ventricle, and mitral valve, offering surgeons a realistic platform to practice patient-specific presurgical simulations.”
“In the past, dynamic heart models have relied on animal tissues to mimic pumping mechanisms. However, this approach raised significant ethical concerns and faced policy restrictions. . . . By combining soft robotics with 3D printing, this research provides a synthetic option to avoid the ethical issues related to animal testing while keeping the physical realism required for medical training. This progress is an important step toward personalized medicine, helping make surgical practice as accurate as possible to improve patient outcomes.” 📰 Full Story →
Intestine-Mimicking Cell Model Accurately Predicts Drug Toxicity
Jeong Chae-bin, The Chosun Daily, 3/5/2026
“A cell model that replicates the structure and function of the human intestine has been developed, creating an evaluation platform capable of more accurately predicting adverse effects of new drugs.”
“The team evaluated the toxicity prediction accuracy of 17 major drugs used in clinical settings, including anticancer agents, targeted therapies, and anti-inflammatory drugs. The results showed a 94% accuracy in predicting gastrointestinal toxicity. Additionally, the model detected early-stage intestinal barrier damage caused by anticancer drugs like paclitaxel, which conventional methods failed to identify, with 92% sensitivity, the team explained.”
“This model is expected to reduce unnecessary clinical failures and evolve into a platform that complements or replaces animal testing.” 📰 Full Story →
New European project develops human-based models for brain disorders
University of Barcelona, News Medical Life Sciences, 3/6/2026
“The University of Barcelona is consolidating its position at the forefront of European research thanks to the strategic project ‘VISI-ON-BRAIN: Cutting-edge Human In Vitro and In Silico Biomedical Tools on Brain Disorders’. This is a training and research programme involving 15 PhD researchers focused on developing new-generation human models to advance the study of complex brain disorders such as Alzheimer’s, Parkinson’s and Huntington’s disease. The key point of the initiative is the paradigm shift it proposes, as the study is carried out leaving behind the animal model and seeking a more ethical science, with experimental (in vitro) and computational (in silico) models.” 📰 Full Story →
TN biotech firm to end all experiments on animals, to use virtual simulation software
Bosco Dominique, The Times of India, 3/6/2026
“Evolute Bioscience, a biotechnology firm . . . has signed an agreement pledging to end all experiments on animals – formerly required in its pharmacology education classes – and instead use virtual simulation software. . . . The decision will save the lives of many animals and fish.”
“ . . . the firm has permanently prohibited the use of living and deceased animals and animal parts in undergraduate and postgraduate pharmacology education, teaching, training, and demonstrations, as well as for research and thesis work in its postgraduate curriculum. Additionally, the firm has permanently canceled its plans to procure at least 50 mice annually for a new educational stream focused on disease model-related animal testing. It will also discontinue using 60 zebra fish for experimental purposes.”
“Simcology’s interactive software, donated by Peta, allows pharmacology studies to conduct experiments using computer-assisted learning models while sparing the lives of animals who otherwise may be forced to inhale or consume chemicals, be infected with diseases, mutilated, and then killed via suffocation or neck dislocation.” 📰 Full Story →
Making a ‘digital twin’ of yourself could revolutionize future surgeries, making medical procedures much more personal
Tia Ghose, LiveScience, 3/8/2026
“[Gastroenterologist] Dr. John Pandolfino . . . [has] begun using ‘digital twins’ to model how individual patients with a swallowing disorder will respond to surgery called myotomy, which is used to cut the esophagus. In the most futuristic conceptions of a digital twin, doctors would combine nearly identical anatomical replicas of an individual’s body with biodata to recommend highly personalized medicine or procedures.”
“‘If you’re looking at surgeries, you wouldn’t need to do this on animals. You would do this on a simulation, like we have, to see what the effects are, and then you could actually go from that to various changes in humans. That’s exactly what happened here: Our virtual esophagus proved what we thought would probably be the right way to do this. So it proved our hypothesis mathematically, and now we’re embarking on a human trial. . . . [Digital twin technology will] take us away from using animals for surgery.” 📰 Full Story →
Growing interest in non-animal respiratory allergy testing – GARD®air data presented by industry leaders
Trading View, 3/9/2026
“Industrial interest in modern, non-animal methods for identifying respiratory allergens is growing, and the field is entering a new phase of increased maturity and relevance. At the world’s largest toxicology conference later this month, the US government’s NICEATM and the global pharmaceutical company Merck will present their independent evaluations of SenzaGen’s GARD®air test method. Together, these presentations indicate that respiratory allergy is emerging as an area of growing interest in non-animal testing approaches.”
“GARD®air is a unique, non-animal method that uses genomics and machine learning to assess whether chemicals and other substances may cause respiratory sensitization. The method is the first of its kind and is suitable for safety assessments in research and development within the chemicals, biotech, and pharmaceutical industries, as well as for evaluating production environments.” 📰 Full Story →
Reducing drug failures with AI, human liver organoids
Kate Barnes, University of Michigan, 3/10/2026
“In conventional drug discovery, nine out of 10 drugs that enter human trials fail for two reasons: The drug does not produce durable effects, or it causes unanticipated side effects and toxicity. Drug-induced liver injury remains one of the leading causes of human clinical trial failures, with more than 20% of promising medications failing due to toxicity issues not detected by traditional animal testing.”
“Through a collaborative national project that includes new experimental and computational technologies being developed at the University of Michigan, researchers aim to reduce this failure rate and improve the drug safety evaluation process, ultimately revolutionizing how drugs are developed. . . . This national project [using human liver organoids and similar heart models combined with predictive AI and physiology-based mathematical models] . . . ‘represents a fundamental shift in how we evaluate drug safety,’ . . . ‘By using human liver organoids derived from diverse patient populations, we can test potential medications in systems that actually reflect human biology.’”
“The models built and trained by the U-M team can predict liver toxicity risk with approximately 90% accuracy. Previous traditional methods achieved closer to 50% accuracy and took months instead of days. This all happens before a drug moves forward to costly clinical trials. This earlier and more accurate testing can improve processes at every stage of drug development, provide safer drugs and potentially drive the cost of medicines down by reducing toxicity prediction failures. The success of these models will also reduce the need for animal testing, which often fails to predict human responses.” 📰 Full Story →
UM researchers involved in landmark study for more ethical, cost-effective way to test protein digestibility
University of Manitoba, 3/16/2026
“As global food systems shift toward sustainability, interest in plant-based, fermented, and alternative proteins is skyrocketing. However, current North American regulations still largely rely on data derived from animal testing. University of Manitoba researchers co-authored and were involved in a major international study that could reduce the food industry’s reliance on animal testing.”
“Protein digestibility is a key factor in determining the nutritional quality of foods, as it reflects how effectively the body can utilize amino acids from dietary proteins. Historically, digestibility values used for food labeling have relied on in-vivo rodent bioassays. While scientifically established, these tests are costly, time-consuming, and raise ethical concerns.”
“The study, supported by the Institute for the Advancement of Food and Nutrition Sciences (IAFNS), involved nine laboratories across Canada and the United States. They evaluated two standardized in-vitro methods (pH-drop and pH-stat assays) for assessing protein digestibility . . . Participating laboratories analyzed the same set of 12 protein ingredients from a variety of plant and animal sources to determine how consistently the tests measured digestibility. The findings demonstrated strong consistency between laboratories and provided scientific support for these methods as practical alternatives to traditional animal-based testing.” 📰 Full Story →
FDA, NIH pledge more flexibility and $150M investment for animal testing alternatives
Darren Incorvaia, Fierce Biotech, 3/18/2026
“Health authorities in the U.S. are continuing their move away from animal testing in drug development, with the FDA signaling an openness to new approaches that haven’t been validated and the National Institutes of Health (NIH) pledging a further $150 million to animal alternatives.”
“The FDA’s Center for Drug Evaluation and Research (CDER) released new draft guidance March 18 specifying that drug approval applicants are welcome to submit data from new approach methodologies (NAMs) like organoids, organs-on-chips and computational models, even if the method hasn’t previously been OK’d by the agency to replace animals. . . . ‘CDER is making it clear that we are hoping to receive appropriate NAMs data in place of animal data when available, as these data can be much more predictive and also a more ethical option,’ the official added. . . . ‘This draft guidance advances our commitment to replace animal testing with human-relevant, scientifically rigorous methods,’ Department of Health and Human Services Secretary Robert F. Kennedy Jr. said in a March 18 release.”
“Over at the NIH, the first awards from the Complement Animal Research In Experimentation (Complement-ARIE) program have been announced, totaling $150 million. This comes on top of the NIH’s previously announced $87 million center for organoid development and a new office leading the charge on NAMs. Through the new awards, the organization is ‘supporting technology development centers and a NAMs data hub and coordinating center,’ the senior NIH official said on the call. The seven new technology centers, per a March 18 release, are ‘designed to stimulate the development of NAMs to address areas of greatest both scientific and regulatory need, with emphasis on biological complexity, throughput and data sharing.’” 📰 Full Story →
$25 Million Project Seeks to Coordinate Data Produced by Human-Based Testing Methods
NYU Langone Health, 3/18/2026
“A research team at NYU Langone Health and Sage Bionetworks has been awarded a $25 million grant to establish the data hub and coordinating center for the National Institutes of Health’s Complement-Animal Research in Experimentation (Complement-ARIE) program. The mission of Complement-ARIE is to speed the development of new approach methodologies (NAMs). These lab- or computer-based testing approaches can more accurately model human biology and complement or replace traditional animal research models.”
“The NIH award to NYU Grossman School of Medicine and Sage is $5 million a year for five years, for a total of $25 million. The award will establish the NYU-Sage NAMs Data Hub and Coordination Center (NYU-Sage NDHCC), which will enable the standardization, harmonization, and sharing of datasets—from molecular tests to 3D cultures to simulated population outcomes—using a cloud architecture. The hub will serve as the consortium’s backbone for data, metadata, code, and computational models; provide AI-augmented data curation and a framework for harmonizing NAMs data across the consortium; and be a source of analytical tools to help researchers work with datasets. The NYU-Sage NDHCC will also foster collaboration among all components of the Complement-ARIE consortium through interactive workshops, benchmarking competitions for an extended community of researchers, and engagement with constituencies vested in the advances of NAMs technologies.” 📰 Full Story →
Texas A&M University launches new NIH-funded center to strengthen chemical safety assessments, reduce animal use
Texas A&M University College of Veterinary Medicine and Biomedical Sciences, Texas A&M University, 3/18/2026
“The Texas A&M University College of Veterinary Medicine and Biomedical Sciences (VMBS), Texas A&M AgriLife Research and partner institutes from across the country have received a five-year, $15.3 million National Institutes of Health (NIH) grant to fund a new research center that aims to transform how industrial and consumer-use chemicals are evaluated for human safety. Funded through the NIH’s Common Fund Complement-Animal Research in Experimentation (Complement-ARIE) program, the New Approach Methodologies (NAMs) Decision Center will advance modern, science-based approaches that improve and accelerate chemical safety assessments while reducing the need for new animal testing.”
“The NAMs Decisions Center brings together a multidisciplinary team of toxicologists, engineers, data analysis experts, regulatory professionals and educators to integrate NAMs — advanced, human based laboratory models and computer-based tools — into real-world regulatory decision-making. . . . Together, these scientists will address barriers that have limited regulatory acceptance of non-animal methods, particularly for non-pharmaceutical chemicals used in everyday products and industrial applications.” 📰 Full Story →
Johns Hopkins Researchers Awarded $15 Million to Develop Platform to Study Neurological Diseases and Screen Chemicals Using ‘New Approach Methodologies’
Johns Hopkins Bloomberg School of Public Health, 3/18/2026
“A team of Johns Hopkins researchers has been awarded a five-year $15 million grant from the National Institutes of Health to develop a platform to test potential new treatments for neurological diseases such as Alzheimer’s, and to screen for harmful chemicals. The grant is through NIH’s Common Fund’s Complement Animal Research In Experimentation (Complement-ARIE) program which supports developing New Approach Methodologies (NAMs) that simulate human biology—complementing and, in some instances, replacing animal testing. This first round of funding will support a series of technology development centers—including one at Johns Hopkins—as well as a NAMs data hub and coordinating center. Total NIH funding for these initiatives is $150 million over five years, pending availability of funds.”
“The Drug Research Organoid Intelligence Development Platform (DROIDp) will use brain organoids—lab-grown neural tissues derived from human stem cells—advanced electrical sensors, and AI analytics to assess neural functions such as learning and memory in drug and chemical testing, and to develop integrated NAMs. . . . ‘The DROID platform is designed to help close one of the important gaps in in vitro neurobiology models: the ability to measure higher-order neural responses such as learning-related activity and memory in a human-relevant system,’ . . . ‘Through the NIH Common Fund’s Complement-ARIE program, we have an opportunity to develop integrated NAMs into a practical framework that can reduce reliance on animal studies.’” 📰 Full Story →
A Billionaire-Backed Startup Wants to Grow ‘Organ Sacks’ to Replace Animal Testing
Emily Mullin, WIRED, 3/23/2026
“As the Trump administration phases out the use of animal experimentation across the federal government, a biotech startup has a bold idea for an alternative to animal testing: nonsentient ‘organ sacks.’ Bay Area-based R3 Bio has been quietly pitching the idea to investors and in industry publications as a way to replace lab animals without the ethical issues that come with living organisms. That’s because these structures would contain all of the typical organs—except a brain, rendering them unable to think or feel pain. The company’s long-term goal, cofounder Alice Gilman says, is to make human versions that could be used as a source of tissues and organs for people who need them.”
“Organ sacks would in theory offer advantages over existing organs-on-chips or tissue models, which lack the full complexity of whole organs, including blood vessels. . . . R3’s ambitions go beyond replacing animal testing, though. The company is eyeing replacement of human parts, an emerging idea in the longevity field. The startup aims to create nonsentient human organ sacks that could provide blood, tissue, and organs to people when their own bodies fail them.” 📰 Full Story →
Organoids: new Roche research centre in Basel will focus on alternative model systems
European Biotechnology, 3/25/2026
“The pharmaceutical group Roche is further expanding its research infrastructure in Switzerland, with a clear focus on novel biological model systems. In Basel, the company has opened a new building for the Institute of Human Biology (IHB), which is set to play a central role in drug discovery. . . . The IHB focuses on so-called human model systems, particularly organoids—lab-grown mini-organs that replicate key functions of human tissues. These are complemented by organ-on-chip technologies and computational models. The aim is to simulate disease processes more realistically and to better predict the effects of new therapies at an early stage.”
“This approach is also intended to reduce reliance on animal testing, which regulators such as the U.S. Food and Drug Administration no longer regard as fundamentally sufficient on its own to model human biology at the level achievable with validated organoid systems. At the same time, it addresses ongoing concerns about the continued high number of laboratory animals used in medical research.” 📰 Full Story →
Human Microphysiological Systems Accelerate Anti-Aging Research
Scienmag, 3/25/2026
“In a transformative advancement poised to revolutionize aging research, scientists have engineered human microphysiological systems (MPS) that authentically recapitulate the natural in vivo aging process. This pioneering work . . . provides an unprecedented platform that accelerates the evaluation of anti-geronic strategies—therapeutics aimed at slowing, halting, or even reversing the cellular and tissue decline associated with aging.”
“Aging is a highly complex, multifactorial phenomenon characterized by the gradual deterioration of physiological functions at cellular, tissue, and systemic levels. Conventional aging studies have relied heavily on animal models or simplistic in vitro systems, both of which present significant limitations. Animal models can fail to fully replicate human-specific aging pathways due to species differences….”
“Importantly, these systems address a crucial ethical consideration by reducing dependence on animal experimentation in aging research.” 📰 Full Story →
British drug regulator plans framework to reduce animal testing reliance
Reuters, 3/25/2026
“Britain’s medicines regulator said on Wednesday it will introduce a framework by the end of 2026, which would allow drugmakers to ask a [sic] review of the data for drugs being developed without animal testing before applying for approval.”
“The draft guidance from the UK’s Medicines and Healthcare products Regulatory Agency aims to reduce reliance on animal studies and aligns with a broader global push to limit such testing in drug development.” 📰 Full Story →
EMA consults on virtual control groups to help reduce animal use in medicines development
European Medicines Agency, 3/31/2026
“EMA’s human medicines committee (CHMP) has issued a draft qualification opinion for a new methodology in preclinical research, which can reduce the overall number of animals (rats) used in specific dose-range finding studies. This method replaces standard (concurrent) animal control groups with virtual control groups. By qualifying this new approach methodology (NAM), the CHMP can accept evidence generated using virtual control groups (within the defined context of use) as scientifically valid in future medicines applications.”
“While the reduction of animal testing will be gradual, this first qualification opinion for a NAM used in the assessment of toxicity, creates a blueprint for future applications. Potential iterations of the NAM could be qualified for use in toxicological studies where control groups are routinely required. Replacing control groups with ‘virtual animals’ in such studies would have a substantial impact on reducing the total numbers of animals used.” 📰 Full Story →
When you share these stories, you’re helping expose a truth the animal research industry wants to hide: ethical science is better for everyone.
Share this science news compilation on Facebook, X, or Bluesky.