While the animal research industry continues to breed, buy, cage, torture, and kill sentient beings, progressive scientists are busy proving that human-relevant science is not only possible but, in fact, better for us all. Highlights in science from the last month are below.
Bio-Techne Expands 3D Stem Cell and Organoid Culture Portfolio with a Fully Defined Synthetic Alternative
Stock Titan, 2/2/2026
“Bio-Techne Corporation . . . announced the launch of CultrexTM Synthetic Hydrogel, a fully defined synthetic extracellular matrix (ECM) designed to support reproducible and scalable 3D stem cell and organoid research.”
“Cultrex Synthetic Hydrogel is designed to support the use of 3D organoid models across a wide range of applications, including drug screening, toxicology and personalized medicine, reducing the reliance on animal-component derived matrices and supporting the broader utilization of new approach methodologies (NAMs). By avoiding the biological variability inherent in traditional ECMs, the hydrogel helps researchers standardize experimental conditions across studies and laboratories, enabling more consistent and reliable organoid culture.” 📰 Full Story →
CompagOs Explores 3D-Printed Bone Models for Early Bone Disease Detection
Julia S., 3D Natives, 2/3/2026
“CompagOs is dedicated to advancing bone health by helping researchers translate scientific discoveries into real-world applications, and ultimately, better treatments for patients. At the heart of its work are biologically reproducible Bon3OID™ in vitro bone models, produced using bioprinting. According to the company, early use of Bon3OID™-DX has the potential to reduce, delay, or even prevent serious complications associated with bone diseases, including fractures, surgery, radiation therapy, and chronic pain—significantly improving patients’ quality of life.”
“‘At the heart of our technology are Bon3OID™ models – 3D bioprinted bone models made from human stem cells . . . The original goal was to create an in vitro model system that provides a biologically relevant environment for researching bones and bone diseases – without relying on conventional 2D models or animal testing. A decisive breakthrough was achieved when the models were able to replicate the clinical picture of osteogenesis imperfecta (brittle bone disease) for the first time. After realizing that Bon3OID™ models offer versatile applications in both research and clinical settings, we decided to commercialize the technology.’” 📰 Full Story →
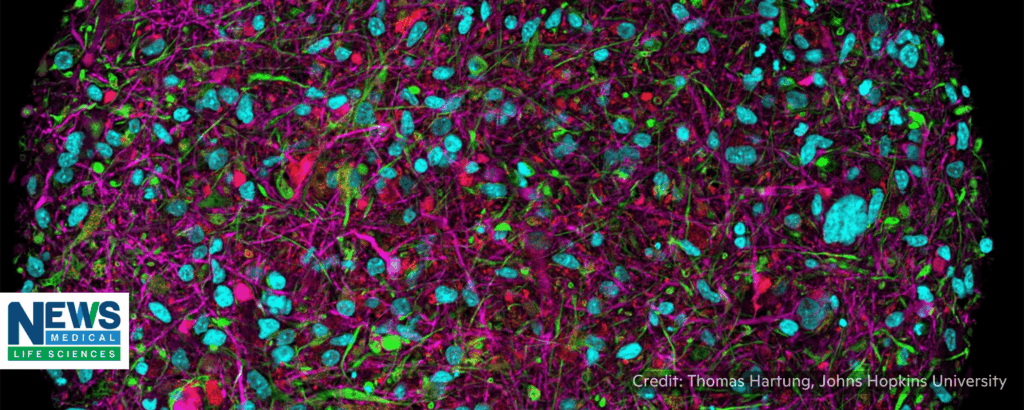
Organoid neurotoxicity testing gains momentum
Pharmaphorum, 2/3/2026
“One in four safety-related failures arise from neurological toxicity, yet, nearly four-fifths of these problems are not recognised until clinical trials, when the risks to volunteers and the financial cost to sponsors are far greater. The shortfall reflects a persistent dependence on models that do not represent human neural biology with sufficient accuracy, leaving a substantial gap between preclinical evidence and clinical outcomes.”
“For decades, most laboratories have relied on animal behaviour studies or simple two-dimensional cell assays to assess neurological risk. Researchers point out that these methods rarely capture the types of functional disturbances that clinicians observe at the bedside . . . As a result, studies frequently miss early signs of risk, while occasionally flagging concerns that later prove irrelevant.”
“Organoid technologies have become a central part of this discussion, particularly for studies involving the central nervous system. Because they are built from human-derived stem cells and develop into multi-cellular structures with a degree of functional organisation, brain organoids provide a way to examine network activity that cannot be replicated with conventional two-dimensional culture. They allow researchers to observe how compounds influence communication between neurons and how disruptions give rise to patterns that resemble early stages of toxicity.” 📰 Full Story →
Tech for good – the death of animal testing? Tech advances might render it obsolete
Madeline Bennett, Diginomica, 2/3/2026
“Many of the medicines in our cupboards and products on our shelves today are tested on animals before they are released to market. But the reality is, most of these tests are a waste of time” and “the vast majority aren’t [required by law].”
“While there’s not always a direct equivalent between non-animal methods and the current ways animals are used, non-animal tests can be combined together to provide very reliable evidence, which is directly relevant to humans. Jessamy Korotoga, Head of Science at Animal Aid, notes: ‘Animal tests are the wrong species and the data from them cannot be reliably translated to humans.’”
“This is where technology has a large part to play in supporting the phase out of animal tests . . . One of these technologies is AcutoX, which was designed by vegan laboratory XCellR8 and part-funded by Animal Aid. AcutoX could be used to replace animals in the oral LD50 toxicity test, which sees live animals being force fed increasing doses of a substance until half of them die. XCellR8’s cruelty-free alternative uses donated human skin cells and looks at the damage caused to these cells by chemicals, instead of using live animals.” 📰 Full Story →
Advancing brain microphysiological systems for in vitro neurotoxicity and cognitive function testing
Yokogawa Life Sciences, News Medical Life Sciences, 2/3/2026
“‘bMPS are 3D models, such as organoids, assembloids, and organ-on-chip platforms, derived from human induced pluripotent stem cells. They aim to replicate the architecture and function of the human brain, capturing key features like network formation and cellular diversity. These models overcome some limitations of 2D cultures and animal models, and so provide more physiologically relevant insights into human neurodevelopment and disease.’”
“‘Animal models are expensive, time-consuming, and often fail to replicate human-specific biological processes. For instance, a single Developmental Neurotoxicity (DNT) study in animals can cost over a million dollars and use more than a thousand rats. bMPS enable higher-throughput testing and allow for studying human-relevant responses, including those linked to sex, genetics, and immune function. They are also better suited for long-term and low-dose exposure studies.’” 📰 Full Story →
Parallel Bio’s [sic] embraces in-house drug development as FDA backs animal testing alternatives
Brian Buntz, R&D World, 2/4/2026
“In 2006, a drug called TGN 1412 met every preclinical safety requirement. Clean in animal models. Clean in monkeys. Ninety minutes after six human volunteers received their first dose at a London hospital, they were fighting for their lives with their immune systems in overdrive. ‘We put it in our system and showed the exact same response,’ says Robert DiFazio, co-founder of Parallel Bio. Its human organoid platform predicted, retrospectively, the failure that animal models missed entirely.”
“Parallel Bio, founded in 2021, builds human immune organoids, functioning in-vitro replicas of lymph nodes that can predict how drugs will perform in people before they reach clinical trials. The company has signed on eight pharmaceutical partners to test drugs using its ‘Clinical Trial in a Dish’ platform. DiFazio and co-founder Juliana Hilliard are now exploring in-house drug development, with an eye toward filing their own INDs . . . The company is building validated disease models across cancer and autoimmune conditions, with plans to expand its portfolio by mid-year.”
“Men are more likely to die from infectious diseases; women are more likely to develop autoimmune conditions, a divergence driven partly by sex hormones that directly regulate immune function. Yet preclinical testing still relies heavily on single-sex inbred mice, collapsing human diversity into a single genetic line. ‘It’s ignoring facts,’ DiFazio says. ‘It’s the reason medicine doesn’t work for everyone.’” 📰 Full Story →
India to swap lab animals for human-on-chip technology. Here’s why
Radifah Kabir, India Today, 2/6/2026
“India’s pharmaceutical sector is at a historic turning point as it moves towards becoming a 500 billion dollar [] global powerhouse by 2047, as part of the country’s Viksit Bharat vision. A landmark report titled ‘Landscape Analysis on Alternatives to Animal Testing for Drug Development in India’ reveals a bold roadmap to swap traditional animal models for cutting-edge, human-relevant technologies.”
“This transition is not just an ethical move but a strategic necessity to strengthen national competitiveness, improve patient outcomes, and foster public trust in modern science.” 📰 Full Story →
Türkiye creates first mini-brain to study rare disease without animal testing
Newsroom, Türkiye Today, 2/7/2026
“Turkish researchers are developing the country’s first laboratory grown mini-brain to study a rare genetic disorder and test future treatments . . . The project will move forward in cooperation with Harvard University. Researchers say the organoid will not only help study [the rare TBL1XR1 genetic disorder] but may also open new research avenues for neurological conditions such as epilepsy and autism.”
“Under the leadership of the Acibadem center, scientists will model a microscopic brain structure that carries the genetic and functional features of the human brain. Alanay said researchers will apply genetic treatments directly to the organoid rather than to patients. ‘This will allow us to test new drugs,’ . . . ‘We will be able to work in such a laboratory environment without moving to animal experiments. We do not want to harm animals, and an experimental animal’s brain can only model the human brain to a limited extent.’” 📰 Full Story →
Gift Seeds Non-Animal Testing Organ-on-a-Chip Initiative at UConn School of Pharmacy
Soibhan Murray, UConn Today, 2/10/2026
“Pharmaceutical leader and philanthropist Jane Hirsh ‘65 (PHARM) made a $250,000 gift to the University of Connecticut’s School of Pharmacy to seed non-animal research in drug development, including organ-on-a-chip innovations.”
“Part of the gift establishes the Jane Hirsh Fellowship in Non-Animal Testing Innovation, which directly supports pharmacy graduate students pursuing alternatives to animal testing in medical therapy development. The balance of the gift will create the Hirsh Fund for Non-Animal Testing Innovation Programs, which funds innovative School of Pharmacy research in non-animal testing methods, including organ-on-a-chip technology. Organ-on-a-chip devices are lined with living human cells and are used to detect human organ responses.”
“‘As biomedical science continues to evolve, it is important that education evolves with it,’ says Hirsh. ‘This initiative is intended to help prepare students to work with modern, human-relevant technologies that can make drug development more effective, more responsible, and ultimately more impactful for patients.’’’ 📰 Full Story →
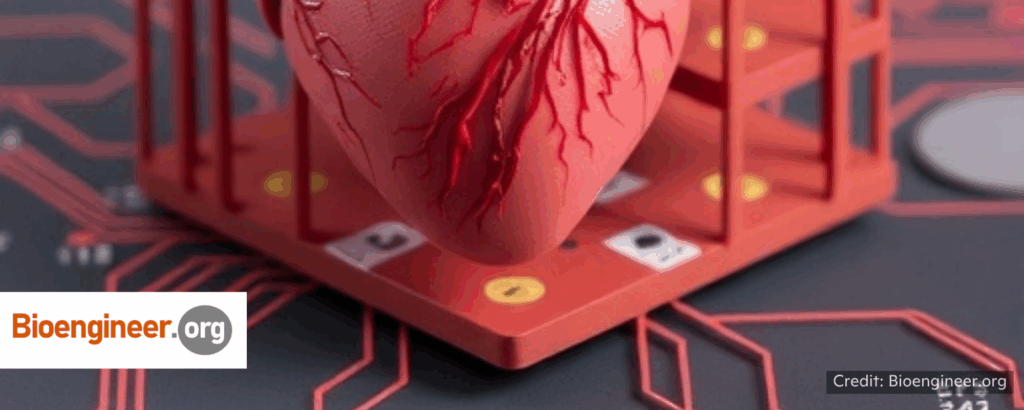
Cardiovascular Disease Models: Heart and Vasculature-on-a-Chip
Bioengineer, Bioengineer.org, 2/12/2026
“The advent of organ-on-a-chip platforms has brought about a transformative shift in the representation of cardiovascular pathophysiology, allowing scientists to delve deeper into the intricacies of disease mechanisms and pharmacological responses across a spectrum of inherited and acquired cardiovascular conditions. Traditional approaches, reliant on static in vitro culture systems and animal models, have frequently fallen short due to their limited biological relevance and the inherent differences that arise from differing species. These shortcomings have stifled the pace of innovation in cardiovascular research, creating an urgent call for more effective models that can accurately mimic human physiology and disease.”
“Heart-on-a-chip and vasculature-on-a-chip are at the forefront of this technological revolution. These sophisticated models boast three-dimensional structures that integrate a variety of cell populations, often sourced from pluripotent stem cells, to create more accurate representations of human cardiovascular tissues. By controlling electromechanical conditions and providing precise biochemical stimuli, these platforms cultivate functional, biomimetic microenvironments that closely resemble the physiological state of human cardiovascular systems. One of the most significant benefits of these models is their ability to recreate the dynamic interactions between various cell types found within the heart and blood vessels under normal and pathological conditions.” 📰 Full Story →
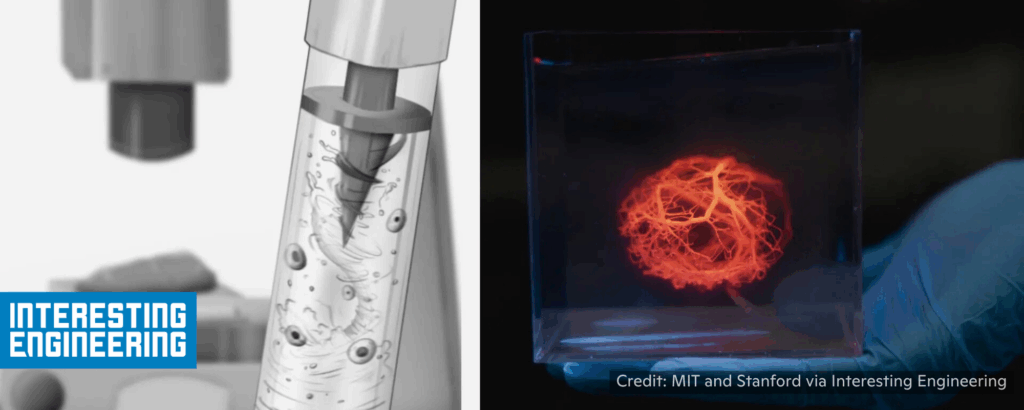
MIT’s new magnetic mixer creates the most uniform 3D-printed tissues ever
Aamir Khollam, Interesting Engineering, 2/10/2026
“Gravity often acts as an enemy in the delicate world of 3D bioprinting. Living cells frequently sink to the bottom of printer syringes. This settling creates clogs and uneven tissue distribution. MIT researchers recently developed a solution called ‘MagMix’ to fix this issue. This compact magnetic mixer keeps bio-inks uniform throughout the entire printing process. Their system allows for the creation of more reliable and consistent human tissues.”
“Consistent bioprinting could eventually reduce the need for animal testing.” 📰 Full Story →
Researchers Developed a New Human-Based Lung Model to Study Highly Infectious Respiratory Illnesses
Physicians Committee for Responsible Medicine, 2/13/2026
“Researchers developed a model of the human alveolus—where air passes from the lungs into blood while breathing—and demonstrated its effectiveness for studying tuberculosis infection . . . Derived from human lung, vascular, and immune cells, this advanced in vitro model replicates key features of alveoli, including the mechanical stretching that occurs during breathing and the air-liquid interface where oxygen enters the blood. Researchers used this model to study early events in tuberculosis infection, but it can also be used to study other lung diseases and test new therapies.”
“Studying tuberculosis in nonhuman animals is inherently limited due to species-specific differences in anatomy and disease progression, pointing to the need for physiologically relevant human-based models of the human alveolus.” 📰 Full Story →
New organoid model helps test spinal cord regeneration drugs
Drug Target Review, 2/17/2026
“Researchers at Northwestern University have created the most advanced organoid model for human spinal cord injury to date. It is hoped that the model will accelerate the development of new therapies for paralysis.”
“In the study, the team used lab-grown human spinal cord organoids to replicate different types of spinal injuries and test a promising regenerative treatment. For the first time, the scientists showed that these organoids could accurately mimic the key effects of spinal cord injury, including cell death, inflammation and glial scarring . . . ‘One of the most exciting aspects of organoids is that we can use them to test new therapies in human tissue,’….” 📰 Full Story →
Revalia Bio’s Quest to Replace Animal Models with Human Organs
Helen Albert, Inside Precision Medicine, 2/18/2026
“Preclinical research has traditionally relied on animal testing as a first step before moving a potential medicine into clinical trials. However, estimates suggest that up to 90% of drug candidates ultimately fail when they reach human trials, primarily due to lack of efficacy or high toxicity, highlighting limitations with animal testing.”
“Revalia’s platform is based on donated human organs rather than animal models, with the aim of making preclinical drug development more human‑relevant, faster, and safer. Many registered organ donors in the U.S. do not end up donating organs for transplantation when they die because diseases such as cancer rule them out. Revalia is aiming to prevent these donations from being wasted and allow the donors to contribute to future research.” 📰 Full Story →
Greenstone Biosciences and Neolaia Bio Announce Strategic Collaboration Using iPSC-Derived Macrophages and Human Gut Organoids to Advance IBD Drug Development
BioSpace, 2/18/2026
“Greenstone Biosciences, a biotechnology company specializing in advanced human disease modeling, New Approach Methods (NAMs), 3D organoids, AI and drug discovery, today announced a strategic collaboration with Neolaia Bio, a biopharmaceutical company targeting the fundamental immune-metabolic drivers of aging biology. Under the collaboration, Neolaia Bio will provide its proprietary drug candidates and discovery expertise, while Greenstone Biosciences will evaluate these compounds using human induced pluripotent stem cell (iPSC)-derived macrophages and patient-relevant gut organoid models. This integrated platform is designed to recapitulate key inflammatory and epithelial features of IBD, enabling more predictive assessment of therapeutic efficacy and mechanism of action.”
“‘This partnership allows us to test our drug candidates in physiologically relevant human systems that closely reflect IBD pathology,’ said Dave Becherer, CSO and Co-founder of Neolaia Bio. ‘By leveraging iPSC-derived immune cells and gut organoids, we aim to generate human-based data that better predicts clinical outcomes and reduces development risk.’ . . . The collaboration reflects a shared commitment to advancing human-relevant preclinical models to address the significant unmet medical needs of patients with IBD.” 📰 Full Story →
New Approach Methodologies Use Nearing The ‘Critical Mass’ Required for Acceptance
Eliza Slawther, Citeline, 2/20/2026
“The use of new approach methodologies (NAMs) within drug R&D activities is playing an increasingly important role for pharma companies, as these animal-free drug testing methods often better translate to human-relevant outcomes than traditional animal tests. This is according to several industry experts….”
“‘I do believe that we’re at a tipping point where we’re seeing a real critical mass converging around these methods that’s really contributing to accelerating their pace of acceptance,’ Holmes [director of science and technology at NC3Rs] said . . . During his presentation, Holmes highlighted the shift towards the validation and acceptance of NAMs by regulators and industry . . . According to Holmes, regulatory and political commitments are beginning to converge with technological advancements to realize the potential of using NAMs in drug development.”
“During the same webinar, experts from AstraZeneca and CN Bio, a UK biotech, spoke about why NAMs tend to produce better results than traditional animal testing when developing certain types of medicine . . . ‘Animals are not humans, and they are becoming increasingly less reliable to predict patient outcomes, especially when testing new modalities, and this is where advanced cell models come into play,’ Cornacchia [Astrazeneca’s director of advanced cell model innovation in safety sciences] said.” 📰 Full Story →
The age of animal experiments is waning. Where will science go next?
Diana Kwon, Nature, 2/25/2026
“Ethical and animal-welfare concerns have long fuelled efforts to curb animal use in research — and now rapid advances in alternative scientific methods are accelerating the shift. These ‘new approach methodologies’ (NAMs) include devices known as organs-on-chips, 3D tissue cultures called organoids and computational models, such as artificial-intelligence systems.”
“Medicines that work in animal models during preclinical testing often prove ineffective in humans. This is one major reason that around 86% of investigational drugs fail in clinical trials, and why many researchers are focused on developing alternatives . . . Increasingly, researchers see NAMs as a way to help. Joseph Wu, a cardiologist and researcher at Stanford University, California, and his team have been developing an approach that they dubbed ‘clinical trials in a dish’. This involves generating induced pluripotent stem cells (iPSCs) from a range of people with a medical condition, using these to grow cells or organoids and then testing whether potential drugs improve how the ‘diseased’ models function.” 📰 Full Story →
New bone marrow model offers scalable platform for next-generation drug discovery
Radcliffe Department of Medicine (University of Oxford), 2/24/2026
“Researchers at the MRC Weatherall Institute of Molecular Medicine have developed the first combined human bone and bone marrow organoid platform capable of modelling long term blood and immune cell production in a fully human 3D system.”
“Despite decades of investment, many therapies for blood cancers and immune disorders fail in late-stage clinical trials due to poor translation from preclinical models. Animal models and simplified lab-grown cell systems often fail to capture the full complexity of human bone marrow biology, limiting their ability to predict what will happen in patients. comBo was developed to address this gap. It integrates bone-forming, supportive and blood-forming cells into a single engineered human micro environment. Within this system, researchers were able to sustain long term human blood cell production (haematopoiesis) and recreate the specialised niches in the bone marrow that regulate stem cell behaviour and immune cell production. . . . By providing a human platform that captures the complexity of bone marrow biology, comBO offers a new foundation for studying blood cancers, immune disorders and regenerative processes. This has the potential to reduce reliance on animal models and accelerate therapeutic discovery.” 📰 Full Story →
University of Sheffield company fighting against animal testing turns two
Camilla Sechi, The Star, 2/24/2026
“Sansanima was first created in 2024, with the aim to find an alternative to animal testing and encourage other companies to adopt this practice. They are currently using their research to test tetanus and botulinum vaccines . . . These vaccines are usually tested on guinea pigs to make sure the strand of tetanus or botulinum are fully deactivated before it is administered to future patients. While testing vaccines is an essential part of their development, these types of tests are categorised as causing the highest level of suffering in animals and even their death.”
“ . . . the company is partnered with the university and both are committed to moving away from practicing animal testing. Sansanima in particular has developed a cell model, known as an assay, which replicates the ‘biology of the tetanus or botulinum toxins’ without needing to test on an animal. The company is sending out this technology to a number of clients to encourage them to adopt it in their research. The technique is not only saving animals from being tested on, but allows medicines to be tested faster.” 📰 Full Story →
Biotech innovator advances life-saving treatments with 3D printing
University of Victoria, 2/25/2026
“[Karolina] Valente, chief executive officer and chief scientific officer of VoxCell BioInnovation is receiving an Emerging Alumni Award from the University of Victoria (UVic) for her leadership in cancer research and drug testing.”
“Shortly after completing her PhD at UVic, Valente founded VoxCell to create 3D bio-printed tissue for drug testing. The company is working to replace animal testing with more accurate, human-relevant models, creating faster, safer and more effective pathways to life-saving treatments. Under Valente’s leadership, the company has raised millions in funding, filed multiple patents, expanded internationally and gained global recognition for its work in oncology and bioprinting.” 📰 Full Story →
When you share these stories, you’re helping expose a truth the animal research industry wants to hide: ethical science is better for everyone.
Share this science news compilation on Facebook, X, or Bluesky.